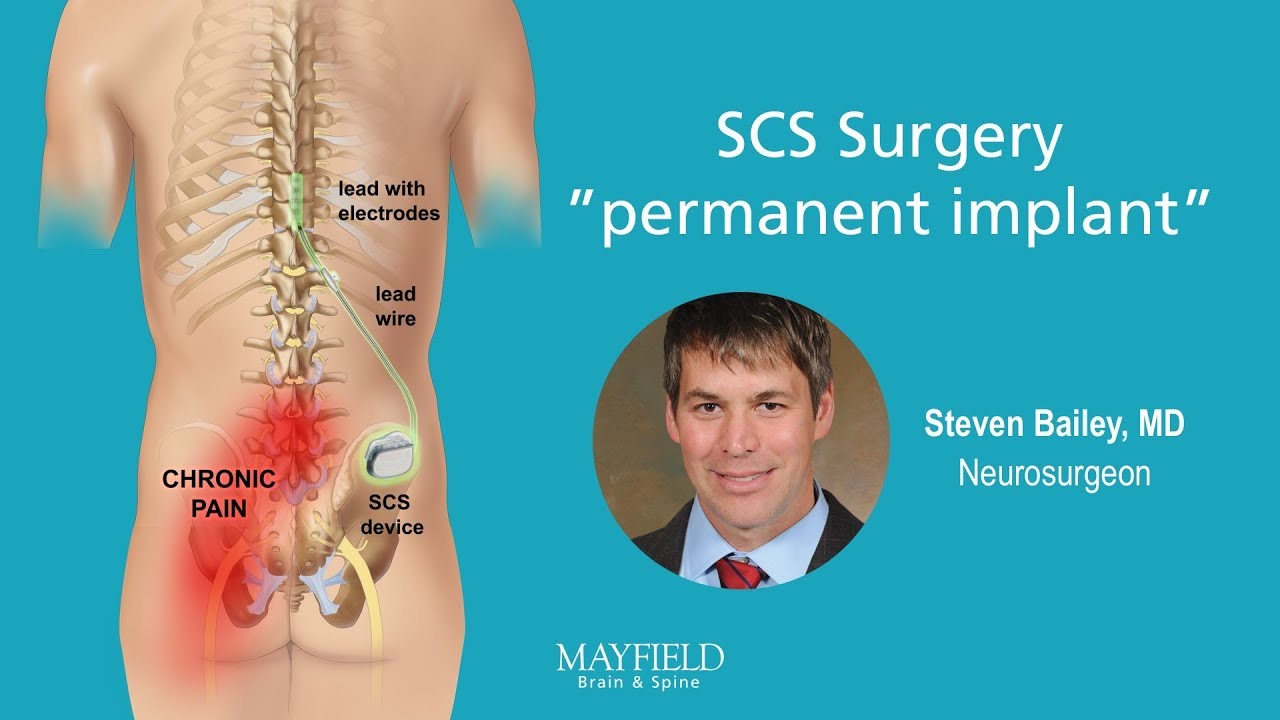
Comparison of conventional, burst and high-frequency spinal cord stimulation on pain relief in refractory failed back surgery syndrome patients: study protocol for a prospective randomized double-blinded cross-over trial (MULTIWAVE study) | Trials
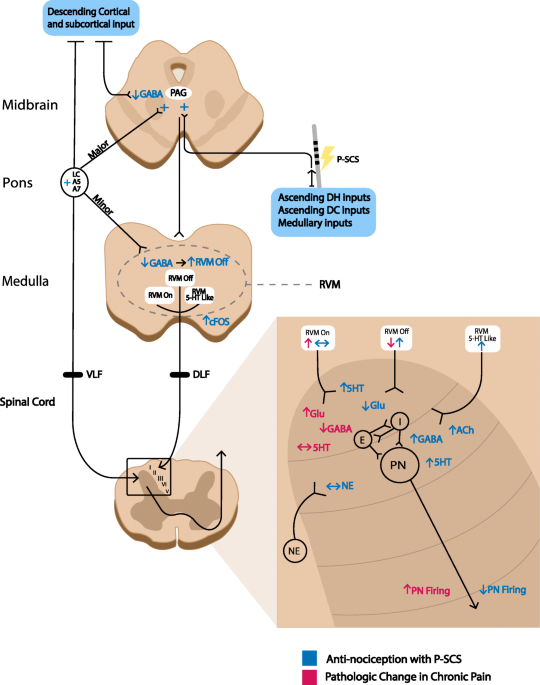
Spinal cord stimulation in chronic pain: evidence and theory for mechanisms of action | Bioelectronic Medicine | Full Text

Nevro Announces FDA Approval of its 10 kHz High Frequency Spinal Cord Stimulation Therapy for Treatment of Chronic Pain Associated with Painful Diabetic Neuropathy (PDN)
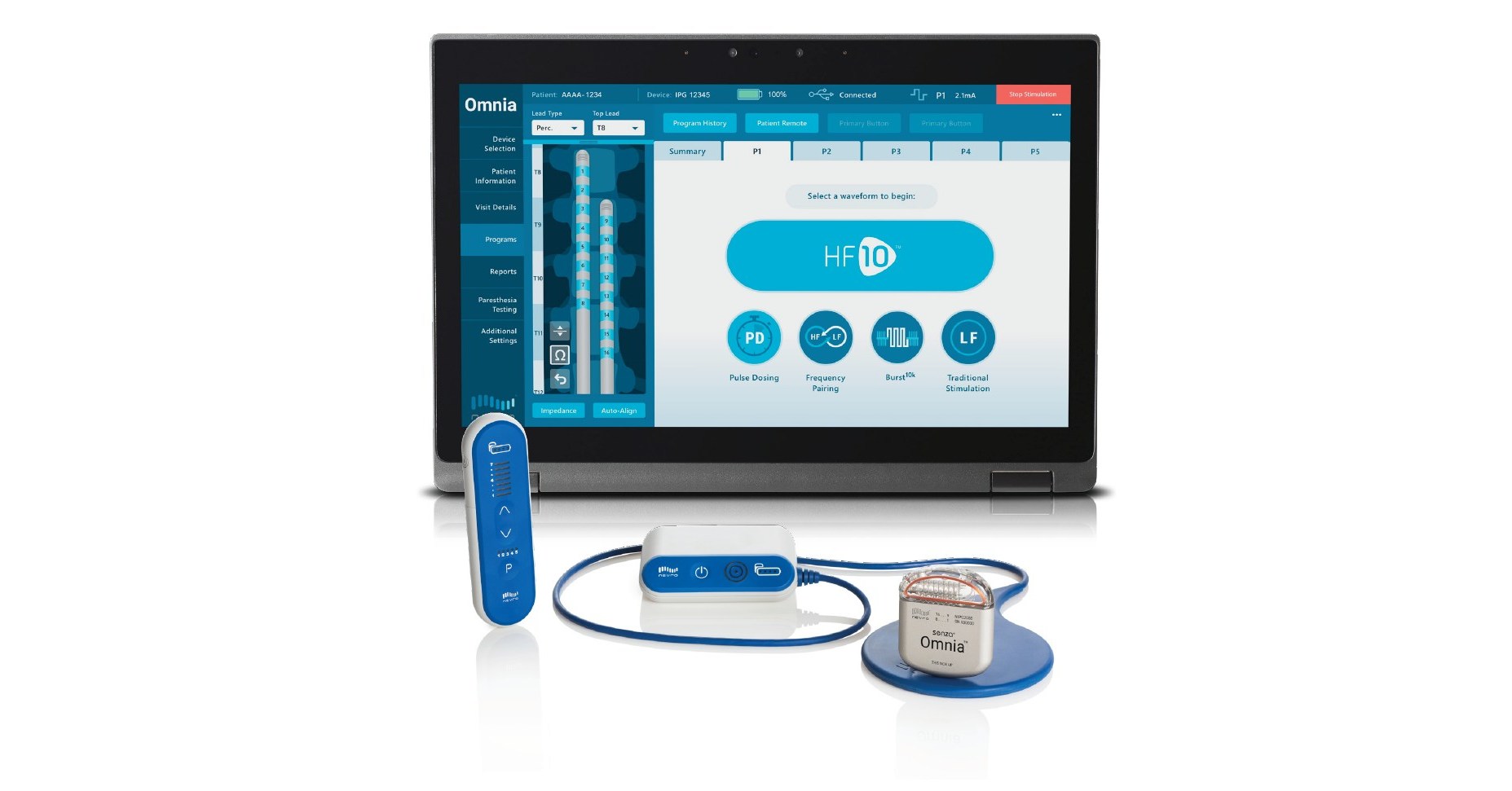
Nevro Announces CE Mark Approval of Senza® Omnia™ Spinal Cord Stimulation System to Treat Chronic Pain

A multicenter real‐world review of 10 kHz SCS outcomes for treatment of chronic trunk and/or limb pain - Stauss - 2019 - Annals of Clinical and Translational Neurology - Wiley Online Library